6 results
Pharmacovigilance analysis of the Vigibase on antidepressants-related withdrawal syndrome in adults and adolescents
- C. Gastaldon, G. Schoretsanitis, E. Arzenton, E. Raschi, D. Papola, G. Ostuzzi, U. Moretti, E. Seifritz, J. M. Kane, G. Trifirò, C. Barbui
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, pp. S95-S96
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Antidepressant discontinuation may cause withdrawal syndrome in some cases. However, evidence on this syndrome related to individual antidepressants is limited, as well as about individual risk factors for severe reactions.
ObjectivesTo ascertain whether each individual antidepressant is associated with an increased reporting of withdrawal syndrome as compared with other medications, and to examine clinical risk factors for severe reactions.
MethodsWe conducted a pharmacovigilance study, with a case/non-case design. We included reports of antidepressant-related withdrawal syndrome from the VigiBase, the WHO global database of individual case safety reports of suspected adverse drug reactions. We performed a disproportionality analysis (calculating reporting odds ratio (ROR) and the Bayesian information component (IC)) of reports of antidepressant-related withdrawal syndrome, comparing antidepressants to all other drugs and to buprenorphine (as a positive control). Antidepressants with significant disproportionate reporting were ranked in terms of clinical priority. We compared serious versus non-serious reactions to determine clinical risk factors for severe reactions.
ResultsBased on 31,688 reports of antidepressant-related withdrawal syndrome, we detected a disproportionate reporting for 23 antidepressants. The ROR for antidepressants altogether, compared to all other drugs, was 14.26 (95%CI:14.08-14.45), 17.01 for other antidepressants (95%CI:16.73-17.29), 13.65 for SSRIs (95%CI:13.41-13.90) and 2.8 for tricyclics (95%CI:2.59-3.02). Based on clinical priority ranking, the strongest signals were found for paroxetine, duloxetine, venlafaxine and desvenlafaxine (figure 1), being comparable to buprenorphine. Severe reactions were more frequently reported in males, adolescents, persons with multiple medications, and with longer treatment duration.
Image:
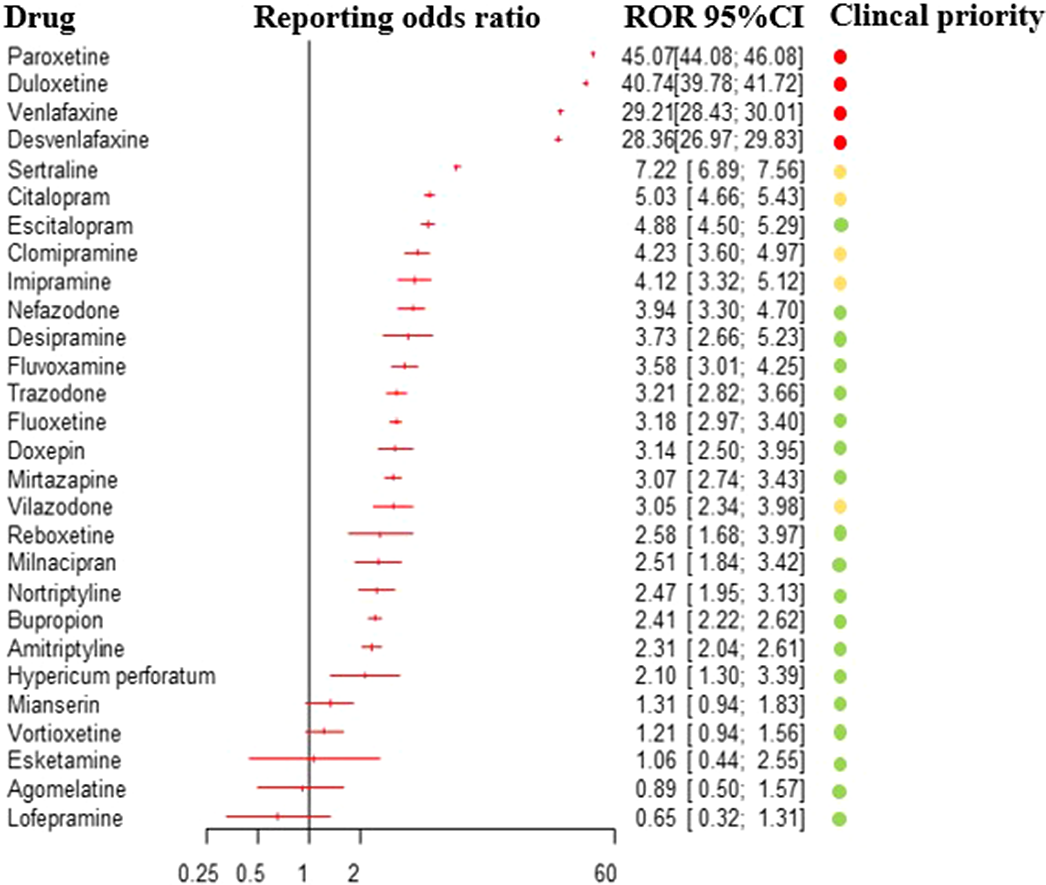 Conclusions
ConclusionsAntidepressants are associated with increased reporting of withdrawal syndrome compared with other medications, with differences between individual antidepressants. Clinicians should be aware of such differences, when prescribing and discontinuing these drugs, as well as of the risk to experience more severe withdrawal symptoms in specific cases.
Disclosure of InterestC. Gastaldon: None Declared, G. Schoretsanitis Consultant of: Dr. Schoretsanitis has served as a consultant for HLS Therapeutics, E. Arzenton : None Declared, E. Raschi : None Declared, D. Papola: None Declared, G. Ostuzzi: None Declared, U. Moretti: None Declared, E. Seifritz Grant / Research support from: Dr. Seifritz has received educational grants, consulting fees and lecture honoraria from Janssen Cilag, Lundbeck, Angelini, Otsuka, Servier, Ricordati, Vifor, Sunovion, Schwabe and Mepha, Consultant of: Dr. Seifritz has received educational grants, consulting fees and lecture honoraria from Janssen Cilag, Lundbeck, Angelini, Otsuka, Servier, Ricordati, Vifor, Sunovion, Schwabe and Mepha, J. Kane Shareolder of: LB Pharmaceuticals and Vanguard Research Group, Consultant of: Dr. Kane has been a consultant and/or advisor for or has received honoraria from Alkermes, Allergan, LB Pharmaceuticals, H. Lundbeck, Intracellular Therapies, Janssen Pharmaceuticals, Johnson and Johnson, Merck, Minerva, Neurocrine, Newron, Otsuka, Pierre Fabre, Reviva, Roche, Sumitomo Dainippon, Sunovion, Takeda, Teva and UpToDate, G. Trifirò Grant / Research support from: he was the scientific director of a Master program on pharmacovigilance, pharmacoepidemiology and real-world evidence which has received non-conditional grant from various pharmaceutical companies; he coordinated a pharmacoepidemiology team at the University of Messina until Oct 2020, which has received funding for conducting observational studies from various pharmaceutical companies (Boehringer Ingelheim, Daichii Sankyo, PTC Pharmaceuticals). He is also scientific coordinator of the academic spin-off “INSPIRE srl” which has received funding for conducting observational studies from contract research organizations (RTI Health Solutions, Pharmo Institute N.V.)., Consultant of: Dr. Trifirò has served in the last three years on advisory boards/seminars funded by SANOFI, Eli Lilly, AstraZeneca, Abbvie, Servier, Mylan, Gilead, Amgen;, Speakers bureau of: Dr. Trifirò has served in the last three years on advisory boards/seminars funded by SANOFI, Eli Lilly, AstraZeneca, Abbvie, Servier, Mylan, Gilead, Amgen;, C. Barbui: None Declared
Pharmacovigilance analysis of the Vigibase on neonatal withdrawal syndrome following in utero exposure to antidepressants
- C. Gastaldon, E. Arzenton, E. Raschi, O. Spigset, D. Papola, G. Ostuzzi, U. Moretti, G. Trifirò, C. Barbui, G. Schoretsanitis
-
- Journal:
- European Psychiatry / Volume 66 / Issue S1 / March 2023
- Published online by Cambridge University Press:
- 19 July 2023, p. S96
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
Evidence on neonatal withdrawal syndrome following antidepressant intrauterine exposure is limited, particularly for antidepressants other than selective serotonin reuptake inhibitors (SSRIs).
ObjectivesTo ascertain whether maternal antidepressant treatment may be associated with withdrawal syndrome in neonates, investigating the comparative reporting between individual antidepressants and classes.
MethodsWe performed a case/non-case pharmacovigilance study, searching reports of withdrawal syndrome in newborns in the VigiBase, the WHO database of suspected adverse drug reactions. Disproportionality analysis was performed, estimating reporting odds ratio (ROR) and the Bayesian information component (IC). Antidepressants were compared to all other medications, to methadone, and within each class of antidepressants (SSRIs, tricyclics (TCA) and other antidepressants). Antidepressants were ranked in terms of clinical priority, based on a semiquantitative score.
ResultsWe retrieved 406 reports of neonatal withdrawal syndrome in 379 neonates related to 15 antidepressants. Compared to all other drugs, disproportionate reporting was detected for antidepressants altogether (ROR: 6.18, 95%CI:5.45-7.01), for TCAs (10.55, 95%CI:8.02-13.88), other antidepressants (ROR: 5.90, 95%CI:4.74-7.36) and SSRIs (ROR: 4.68, 95%CI:4.04-5.42). All antidepressants showed a significant disproportionality, apart from bupropion (figure 1). We did not find any disproportionate reporting for any antidepressant compared to methadone. The clinical priority ranking showed moderate clinical priority for all antidepressants, with the exception four, that had a weak one (figure 1). Most frequently reported symptoms were respiratory symptoms (n=106), irritability/agitation (n=75), tremor (n=52) and feeding problems (n=40).
Image:
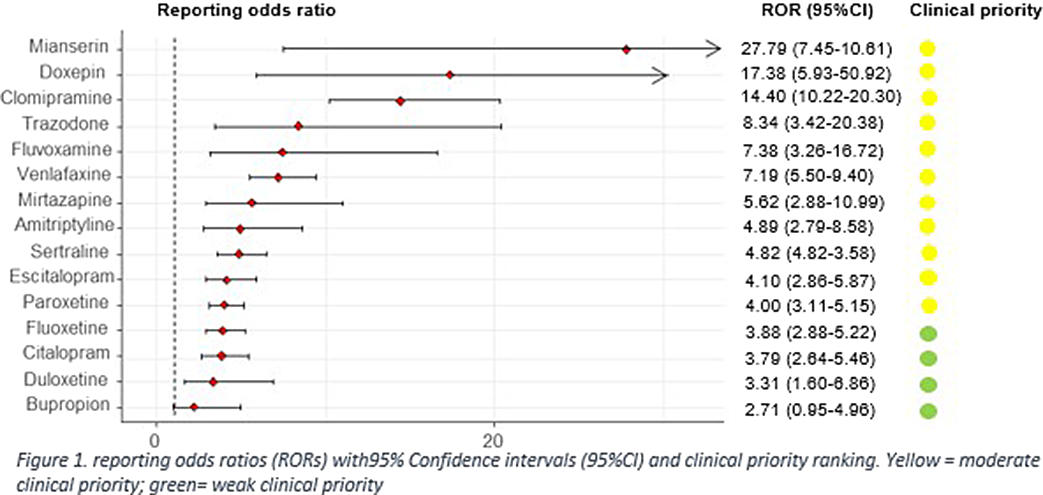 Conclusions
ConclusionsExposure to antidepressants in utero is associated with moderate signals of disproportionate reporting for neonatal withdrawal syndrome for most antidepressants. Clinicians should pay extra attention to neonates with antidepressant-treated mothers.
Disclosure of InterestC. Gastaldon: None Declared, E. Arzenton : None Declared, E. Raschi: None Declared, O. Spigset: None Declared, D. Papola: None Declared, G. Ostuzzi: None Declared, U. Moretti: None Declared, G. Trifirò Grant / Research support from: he was the scientific director of a Master program on pharmacovigilance, pharmacoepidemiology and real-world evidence which has received non-conditional grant from various pharmaceutical companies; he coordinated a pharmacoepidemiology team at the University of Messina until Oct 2020, which has received funding for conducting observational studies from various pharmaceutical companies (Boehringer Ingelheim, Daichii Sankyo, PTC Pharmaceuticals). He is also scientific coordinator of the academic spin-off “INSPIRE srl” which has received funding for conducting observational studies from contract research organizations (RTI Health Solutions, Pharmo Institute N.V.)., Consultant of: has served in the last three years on advisory boards/seminars funded by SANOFI, Eli Lilly, AstraZeneca, Abbvie, Servier, Mylan, Gilead, Amgen, Speakers bureau of: has served in the last three years on advisory boards/seminars funded by SANOFI, Eli Lilly, AstraZeneca, Abbvie, Servier, Mylan, Gilead, Amgen, C. Barbui: None Declared, G. Schoretsanitis Consultant of: Dr. Schoretsanitis has served as a consultant for HLS Therapeutics and Thermo Fisher Scientific.
Neonatal withdrawal syndrome following in utero exposure to antidepressants: a disproportionality analysis of VigiBase, the WHO spontaneous reporting database
- C. Gastaldon, E. Arzenton, E. Raschi, O. Spigset, D. Papola, G. Ostuzzi, U. Moretti, G. Trifirò, C. Barbui, G. Schoretsanitis
-
- Journal:
- Psychological Medicine / Volume 53 / Issue 12 / September 2023
- Published online by Cambridge University Press:
- 21 September 2022, pp. 5645-5653
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background
Evidence on neonatal withdrawal syndrome following antidepressant intrauterine exposure is limited, particularly for antidepressants other than selective serotonin reuptake inhibitor (SSRIs).
MethodsIn our case/non-case pharmacovigilance study, based on VigiBase®, the WHO database of suspected adverse drug reactions, we estimated reporting odds ratio (ROR) and the Bayesian information component (IC) with 95% confidence/credibility intervals (CI) as measures of disproportionate reporting of antidepressant-related neonatal withdrawal syndrome. Antidepressants were first compared to all other medications, then to methadone, and finally within each class of antidepressants: SSRIs, tricyclics (TCA) and other antidepressants. Antidepressants were ranked in terms of clinical priority, based on semiquantitative score ratings. Serious v. non-serious reports were compared.
ResultsA total of 406 reports of neonatal withdrawal syndrome in 379 neonates related to 15 antidepressants were included. Disproportionate reporting was detected for antidepressants as a group as compared to all other drugs (ROR: 6.18, 95% CI 5.45–7.01, IC: 2.07, 95% CI 1.92–2.21). Signals were found for TCAs (10.55, 95% CI 8.02–13.88), followed by other antidepressants (ROR: 5.90, 95% CI 4.74–7.36) and SSRIs (ROR: 4.68, 95% CI 4.04–5.42). Significant disproportionality emerged for all individual antidepressants except for bupropion, whereas no disproportionality for any antidepressant was detected v. methadone. Eleven antidepressants had a moderate clinical priority score and four had a weak one. Most frequent symptoms included respiratory symptoms (n = 106), irritability/agitation (n = 75), tremor (n = 52) and feeding problems (n = 40).
ConclusionsMost antidepressants are associated with moderate signals of disproportionate reporting for neonatal withdrawal syndrome, which should be considered when prescribing an antidepressant during pregnancy, irrespective of class.
Safety of esketamine nasal spray: Analysis of post-marketing reports submitted to the FDA adverse event reporting system in the first year on the market
- C. Gastaldon, E. Raschi, J. Kane, C. Barbui
-
- Journal:
- European Psychiatry / Volume 64 / Issue S1 / April 2021
- Published online by Cambridge University Press:
- 13 August 2021, pp. S150-S151
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Introduction
The approval of the esketamine nasal spray for treatment-resistant depression in March 2019 by the Food and Drug Administration (FDA), and few months later by the European Medicine Agency, triggered a vivid debate and many concerns, mainly because of the lack of convincing evidence on its efficacy and safety, based on the development programs, approval trials and few post-marketing trials.
ObjectivesWe aimed to detect and characterize safety signals for esketamine, by analyzing relevant adverse events (AEs) reports in the FDA Adverse Event Reporting System (FAERS) database (March 2019-March 2020).
MethodsWe performed disproportionality analysis through the case/non-case approach: reporting odds ratios (ROR) and information components (IC) with 95% confidence intervals (95%CI) were estimated for esketamine-related AEs with at least four counts. We compared serious and non-serious AEs using non-parametrical tests.
ResultsThe FAERS database registered 962 reports of esketamine-related AEs in one year. Signals (i.e., statistically significant disproportionality) were detected for several AEs, such as dissociation, sedation, feeling drunk, suicidal ideation and completed suicide. Signals for suicidal ideation, but not suicide attempt and completed suicide, remained significant when comparing esketamine to venlafaxine. The comparison of patients with serious vs. non-serious esketamine AEs revealed that females, patients receiving antidepressant polypharmacy, co-medication with antipsychotics, mood stabilizers, benzodiazepines or somatic medications were more likely to suffer from serious AEs.
ConclusionsThis real-world pharmacovigilance analysis detected signals of serious unexpected esketamine-related AEs, thus reinforcing current worries regarding esketamine safety/acceptability. Further real-world studies are urgently needed to unravel the safety profile of esketamine.
DisclosureNo significant relationships.
Stomatal numbers in holm oak (Quercus ilex L.) leaves grown in naturally and artificially CO2-enriched environments
- Edited by A. Raschi, Institute of Environmental Analysis and Remote Sensing for Agriculture, Florence, F. Miglietta, Institute of Environmental Analysis and Remote Sensing for Agriculture, Florence, R. Tognetti, Institue of Forest Tree Breeding, Florence, P. van Gardingen, University of Edinburgh
-
- Book:
- Plant Responses to Elevated CO2
- Published online:
- 10 February 2010
- Print publication:
- 28 November 1997, pp 197-208
-
- Chapter
- Export citation
-
Summary
SUMMARY
The objects of the present study were:
1) to investigate the stomatal morphology of leaves of holm oak trees grown in a naturally CO2-enriched environment;
2) to compare it with the stomatal density of leaves of holm oak seedlings grown in an artificially CO2-enriched environment.
Among the stomatal morphology parameters we analysed, the only significant alteration we observed in trees which had grown by the CO2 spring was a reduction in stomatal density.
The rather special response of one tree might be related to the rock on which it stands, which may be causing the tree severe water stress. However, the reduction did not increase significantly as the concentrations of CO2 increased.
The experiment in artificially CO2-enriched atmospheres indicated that the stomatal density of holm oak leaves was reduced by an increase in CO2. The results are discussed with an analysis of herbarium holm oak leaves.
INTRODUCTION
The effect on stomatal density of the increase of atmospheric CO2 is not yet fully clarified, although some experiments exposing plants to elevated CO2 regimes in small scale chambers and field enclosures indicate that stomatal density decreases with increasing CO2 concentrations (Madsen, 1973; O'Leary & Knetch, 1981; Thomas & Harvey, 1983; Imai, Coleman & Yanagisawa, 1984; Woodward, 1987), despite contradictory responses which were sometimes observed.
Criticisms have been levelled at this kind of experiment because of poor balance between energy supply and water loss from leaves (Morison, 1987). Conversely, natural vegetation is well coupled with the atmosphere, in terms of both its energy budget and its environmental feedback mechanisms.
10 - Relations between sap velocity and cavitation in broad-leaved trees
-
- By M. Borghetti, Istituto Miglioramento Genetico delle Piante Forestall, Consiglio Nazionale delle Ricerche, via S. Bonaventura 13, 50145 Firenze, Italy; Present address: Dipartimento di Produzione Vegetale, Universita della Basilicata, Potenza, Italy., P. de Angelis, Dipartimento Scienze dell'Ambiente Forestale e delle sue Risorse, Universitá della Tuscia, via S. Camillo De Lellis, 01100 Viterbo, Italy., A. Raschi, Istituto di Analisi Ambientale e Telerilevamento applicati all'Agricoltura, Consiglio Nazionale delle Ricerche, p.le delle Cascine 18, 50144 Firenze, Italy., G. E. Scarascia Mugnozza, Dipartimento Scienze dell'Ambiente Forestale e delle sue Risorse, Universitá della Tuscia, via S. Camillo De Lellis, 01100 Viterbo, Italy., R. Tognetti, Istituto Miglioramento Genetico delle Piante Forestall, Consiglio Nazionale delle Ricerche, via S. Bonaventura 13, 50145 Firenze, Italy., R. Valentini, Dipartimento Scienze dell'Ambiente Forestale e delle sue Risorse, Universitá della Tuscia, via S. Camillo De Lellis, 01100 Viterbo, Italy.
- Edited by M. Borghetti, J. Grace, A. Raschi
-
- Book:
- Water Transport in Plants under Climatic Stress
- Published online:
- 04 August 2010
- Print publication:
- 13 May 1993, pp 114-128
-
- Chapter
- Export citation
-
Summary
SUMMARY
Concurrent measurements of cavitation by the ultrasound acoustic emission technique and sap velocity by the thermoelectric heat pulse method were carried out in the field on three woody species (Quercus pubescens, Quercus ilex and Alnus cordatd) characterized by different wood structure. The plant water status was assessed by measuring xylem water potential and stomatal conductance. A good correspondence was found between the patterns of sap velocity and cavitation rate. A threshold-type relationship was observed, in Alnus cordata, between water flow and cavitation rate. In some cases temporal lags between ultrasound emission and sap velocity were observed: several factors may account for these lags, including the possibility that cavitation of xylem conduits may be a rather patchy phenomenon and that different xylematic volumes might have been sensed by the ultrasound and heat pulse transducers.
INTRODUCTION
The formation and spreading of gaseous emboli through the xylem are recognized as common events in water stressed plants (Milburn, 1979; Tyree & Sperry, 1989a). In particular, it is widely held that the increase of xylematic tension, which is caused by the drop of water potential between the soil and the atmosphere, frequently induces cavitation, i.e. the breakage of water columns and the formation of gas bubbles in the lumina of xylem conduits.
The mechanism of cavitation is still being debated, although the hypothesis that cavitation is caused by the aspiration of air bubbles through the intervessel pit membranes (the so-called air seeding hypothesis) is widely supported by experimental evidence (Sperry & Tyree, 1988; Sperry, Tyree & Donnelly, 1988).





